
To learn more about molecular shapes, you can read about "Valence Shell Electron Pair Repulsion (VSEPR) Theory." If you need to learn how to draw Lewis structures, check out "Lewis Diagrams." Types of Dipole in Chemistry On the other hand, PCl5 is considered non-polar because its symmetrical shape, which is trigonal bipyramidal, cancels the dipoles out. 
If the molecule is perfectly symmetric, all vectors will add up to zero, and the dipole moment of the whole molecule will be zero.įor example, in PCl3, the bond is polar because of the difference in electronegativity between P and Cl atoms, and the presence of a lone pair of electrons gives it a tetrahedral structure. 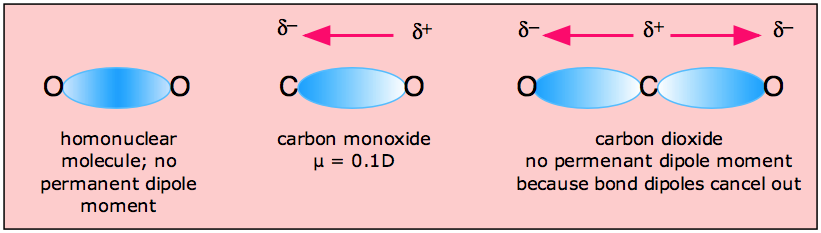
The dipole moment of a molecule is the sum of the dipole moments of the bonds. Dipole moments are measured in Debye units, and the bigger the dipole moment of the bond, the more polar the bond is. The dipole moment of a bond can be calculated using the partial charges and the distance vector between the two charges. Periodic table showing Pauling's scale of electronegativityĭipole moments are a measure of the magnitude of a dipole, and we use arrows pointing towards the more electronegative atom to show it. /chapter3/pages3and4/page3and4_files/dipole.png)
You can see a trend on the periodic table - electronegativity increases from left to right and decreases down a group. The electronegativity values are listed on Pauling's electronegativity scale. If the difference is greater than 1.7, it's an ionic bond. If the difference is between 0.4 and 1.7, it's a polar covalent bond. If the difference is less than 0.4, it's a non-polar covalent bond. To figure out what type of bond is present, we need to look at the difference in electronegativity between the atoms involved. Ionic bonds don't have dipoles, polar covalent bonds always have dipoles, and non-polar covalent bonds have dipoles that cancel out due to symmetry. In polar covalent bonds, electrons are shared unequally, and in ionic bonds, electrons are transferred. In non-polar covalent bonds, electrons are shared equally. There are three types of bonds: non-polar covalent, polar covalent, and ionic. Determination and Formation of a Dipoleĭipoles are formed depending on the polarity of a bond, which is determined by how much an atom can attract electrons to itself - this is called electronegativity. A dipole is a molecule or covalent bond that has a separation of charges. What is the Definition of a Dipole in Chemistry?ĭipoles occur when electrons are shared unequally between atoms in the same molecule due to a high difference in the electronegativity of the atoms involved. So, next time you're drinking a glass of water, remember that it's not just a boring liquid - it's got some seriously cool chemistry going on! And now you know a little more about what makes it so special. This is what makes water stick together and form droplets. For example, there's something called a "hydrogen bond" that happens between water molecules because of their dipoles. There are different types of dipoles in chemistry, but we'll focus on the ones that involve water. This happens because of the way the atoms in water are arranged. In water's case, it has a positive charge on one side and a negative charge on the other. But have you ever heard of water being a "dipole"? If you're curious about what that means, keep reading!īasically, a "dipole" is just a fancy word for something that has two different charges. Water is super cool for lots of reasons! You've probably heard about how it's polar, sticks to things, and dissolves stuff.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
